
Focus Sifter Machine Production
Material: All stainless steel 304
Number of layers: 1-5 layers are optional
Screen specifications: 20-200 mesh
Net cleaning method: bouncing ball + ultrasonic
Applicable standards: Meet GMP requirements
Applicable output: 100-1000kg/h
Chinese medicine powder vibrating screen GMP standard is a high-standard equipment designed for screening Chinese medicine powder. Chinese medicine powder usually has fine particles, high viscosity, easy to absorb moisture and other characteristics, and has extremely high requirements for screening accuracy and hygiene. The equipment uses the principle of three-dimensional vibration to achieve fast and efficient classification, and is suitable for screening 80-500 mesh fine powder. The common processing capacity is between 100-3000kg/h, depending on the material characteristics and mesh size. Because it meets GMP standards, the whole machine is made of stainless steel, the structure is easy to clean, and there is no dead corner, ensuring the safety of drug production. The current market price ranges from about US$2,000 to US$10,000, depending on the specifications and customization requirements.
The GMP (Good Manufacturing Practice) standard has strict requirements for medical equipment, especially emphasizing the cleanliness, material safety and maintainability of the equipment. For the vibrating screen of Chinese medicine powder, the GMP standard is mainly reflected in the following aspects:
Material requirements: The part in contact with the material must be made of 304 or 316L food-grade stainless steel to ensure that it does not react with the powder.
Structural design: No dead angle, can be quickly disassembled, easy to clean and disinfect, and prevent cross contamination.
Sealing performance: Equipped with a high-sealing silicone ring to prevent dust leakage and keep the production environment clean.
Stable operation: The amplitude of the sieve machine is adjustable, and the operation is stable to ensure continuous and efficient screening.
Recording and verification: The equipment must comply with the verification specifications to facilitate the recording and traceability of the production process.

Common parameter configurations are as follows:
Screening mesh: 80 mesh ~ 500 mesh (customizable)
Number of layers: 1~3 layers (supports multi-stage screening)
Processing capacity: 100kg/h ~ 3000kg/h
Power: 0.25kW ~ 1.5kW (depending on the model)
Screen material: 304/316L stainless steel, tensioning or ultrasonic device optional
Equipment diameter: 400mm ~ 1500mm
Material of the whole machine: all stainless steel design, polishing treatment Ra<0.6μm
Voltage: 220V/380V, 50/60Hz
Noise level: <75dB

Common models on the market are divided by diameter and number of layers. Some typical models are as follows:
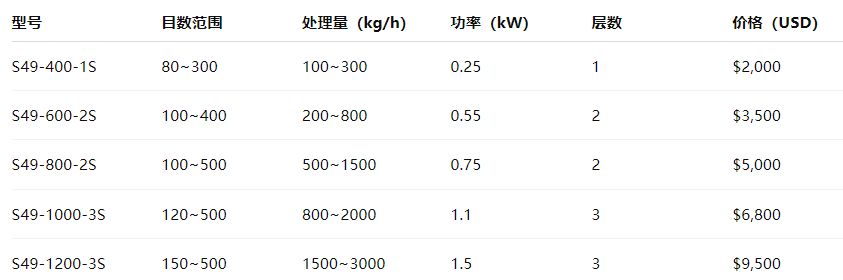
Description: S49 is a commonly used vibrating screen series. The number after the model represents the diameter of the screen machine, and S is the number of layers. The specific parameters can be customized according to needs.
When selecting, factors such as material properties, screening accuracy, output requirements and plant layout should be considered comprehensively:
Material properties: If the Chinese medicine powder contains more fibers or is easy to agglomerate, you can consider installing an ultrasonic system to improve the screening efficiency.
Mesh requirements: Select a sieve with a suitable mesh number according to the particle size requirements of the finished product to ensure the purity and particle consistency of the medicine powder.
Production matching: It is recommended to select a model with a daily processing volume of more than 1000mm, and a multi-layer structure to improve the screening efficiency.
Cleaning and maintenance: All stainless steel and quick-disassembly structures are preferred to meet GMP cleaning verification requirements and reduce downtime for cleaning.
Budget control: The basic model can already meet conventional screening needs. If ultrasonic, airtight, vacuum, etc. are required, the configuration can be gradually upgraded.